Host manipulation induced by parasites is broadly distributed in nature and it has been a topic of several studies over the past decades (Thomas et al. 2010). Ophiocordyceps unilateralis (Clavicipitaceae: Hypocreales) is a fungal pathogen-specific to ants of the tribe Camponotini (Formicinae: Formicidae) with global distribution. Among these myrmecophilous species, this so-called zombie or brain-manipulating fungus alters the behavior of the ant host, causing it to die in an exposed position, typically clinging onto and biting into the adaxial surface of midribs of shrub leaves by its mandibles often followed by the growth of hyphae. Species within this group exhibit Hymenostilbe asexual morphs with the ascospores actively shooting out spores.

Ophiocordyceps (Ophiocordycipitaceae, Hypocreales, Ascomycota) is known as the largest genus of entomopathogenic fungi with around 240 defined species (Robert et al. 2005, 2013); Ophiocordyceps members do not form resting spores (i.e., chlamydospores) and cannot survive outside of their hosts.
Occurrence
Infections of ants by entomopathogenic fungi have been reported in several regions of the world especially in tropical forests, with relatively few reports from temperate ecosystems, such as Asia, Australasia, Africa, and the Americas (Pontoppidan et al. 2009), and diversity concentrated in the tropics and decreasing with increasing latitude. These interactions frequently happen as epizootics, contaminating a large number of ant specimens in small forest regions (Andersen et al. 2009, Pontoppidan et al. 2009). The probability of affecting directly and indirectly on the abundance of its hosts. Among these associations, it is far regarded that Ophiocordyceps species (Hypocreales: Ophiocordycipitaceae) can parasitize numerous ant species from basal groups (e.g. Ponerines) to modern genera (e.g. Camponotus) (Evans & Samson 1982, Sanjuan et al. 2011, Evans et al. 2011). In addition, the occurrence of parasitized ants is fixed upon specific host plants, plant structures, and heights above the soil. In the southeast, Evans et al. (2011) interpreted four new species with conspicuous different microscopic characteristics, but with morphology just like that of O. unilateralis. After this current discovery, minute research was made on the ecology and distribution of these fungi species.
The Fungal Infection Cycle and Host Specificity
The parasitic fungi such as the host-specific pathogen Ophiocordyceps unilateralis sensu lato can dramatically monitor insect brains and manipulate their behavior to arrive death sites that are optimal for spore dispersal; for example, crickets or grasshoppers commit suicide by drowning when infected by the parasitic hairworm Spinochordodes tellinii the so-called fungal extended phenotype (The behavior, in which genes of the parasite are expressed in the host’s phenotype (Dawkins 1982)). On the other hand, on top of physiological immune surveillance, insects (especially social insects such as ants and termites) can smell and eliminate fungal pathogens, groom each other to clear pathogenic spores, raise a fever response, or die well away from their nestmates, that is termed behavioral or social immunity. Thus colony transmission and fungal development are limited by strong social immunity demonstrated by social insect societies.

The Fungal Infection Cycle and Host Specificity Entomopathogenic fungi recognize and infect insects through the spore adhesion and formation of appressoria by attaching, germinating, and penetrating the cuticles of foraging ant workers. After reaching the hemocoel (body cavity) of an insect, fungal filaments will convert into yeast-like cells as single cells in the host hemolymph that go through budding for quick propagation and check the immune response of the hosts.
The infected, moribund ants basically behave like zombies; they walk alone and erratically climb to a definite height in the vegetation (approximately 25 cm above the soil surface), before being killed for the post mortem fungal growth. They bite onto substrate in rainforests and twigs in temperate woods and transition from wandering to biting takes place synchronously around noontime (within 11:00–14:00 h) possibly in association with a solar cue. Before mummifying its host, the fungus drives a larva close to the surface of the soil (approximately 1–3 cm), and then the fruiting body then erupts from its head to form a branching network cadaver of cells. Infections by obligate Entomophthorales pathogens could also lead to similar summit” diseases, i.e., the infected insects climbing to an elevated position before death. Such manipulation functions benefit the fungi by maximizing spore transmission efficiency to begin the next infection cycle.

The fresh stage represents the preliminary phase of fungal development in the ant, which denotes that the host had been recently killed (about 5 days before). The immature stage refers to specimens lacking ascomata but presenting stromata. The rise of compact hyphae from the host’s dorsal pronotum constructing evident stromata (stalks) is characteristic of this group of fungi. Ascomata (fertile parts) initiate on these stromata and disclose the sexual spores that are ejected and spread out after they arrive at their maturity(Kepler et al. 2011; Evans et al. 2011b). Thus the fungus ends the parasitic life cycle by lysing the host tissue. During the ~ 1–2 weeks in which hyphal development occurs in the ant, the fungus is under selective pressure for host resource exploitation and defense against invaders.
Control of Insect Behavior by Parasitic Fungi
Generally, the manipulated behaviors include convulsion, erratic walking. The ants adhered to the plants using their mandibles, normally on the abaxial surface and central veins of leaves, a behavior which is not apparent in healthy ants of this species. According to Andersen et al. (2009), this behavior is congenital to the fungus, ensuring a stable microclimatic niche for its development after the host’s death and facilitating aerial dispersion of the spores. Species belonging to O. unilateralis complex initiate a series of synchronized events within the host to induce it to leave the colony towards the understory, where it perishes in an elevated and strategic position, characteristically attaching itself to abaxial vein or edges of leaves or twigs in a “death-grip” and move and die away from the nest. Once there, the spore-producing structure (stroma) emerges from the anterior intersegmental region to the pronotum, from which the ascospores actively liberate their spores after a few weeks of maturation (Andersen et al. 2009, Araújo et al. 2015). After releasing, the spores are deposited on the forest floor, where they will infect other ants that eventually go on through the site. Invasion of the host body appears through the cuticle and the corpses are predominantly observed in areas where the density of ants previously manipulated and killed by the fungus is already quite intense, called graveyards (Pontoppidan et al. 2009). In Brazil, natural epizootics resulting due to the fungi O. unilateralis were demonstrated in adult individuals of ants Camponotus sp. in the Amazon Forest (Andrade 1980, Evans & Samson 1982, 1984).
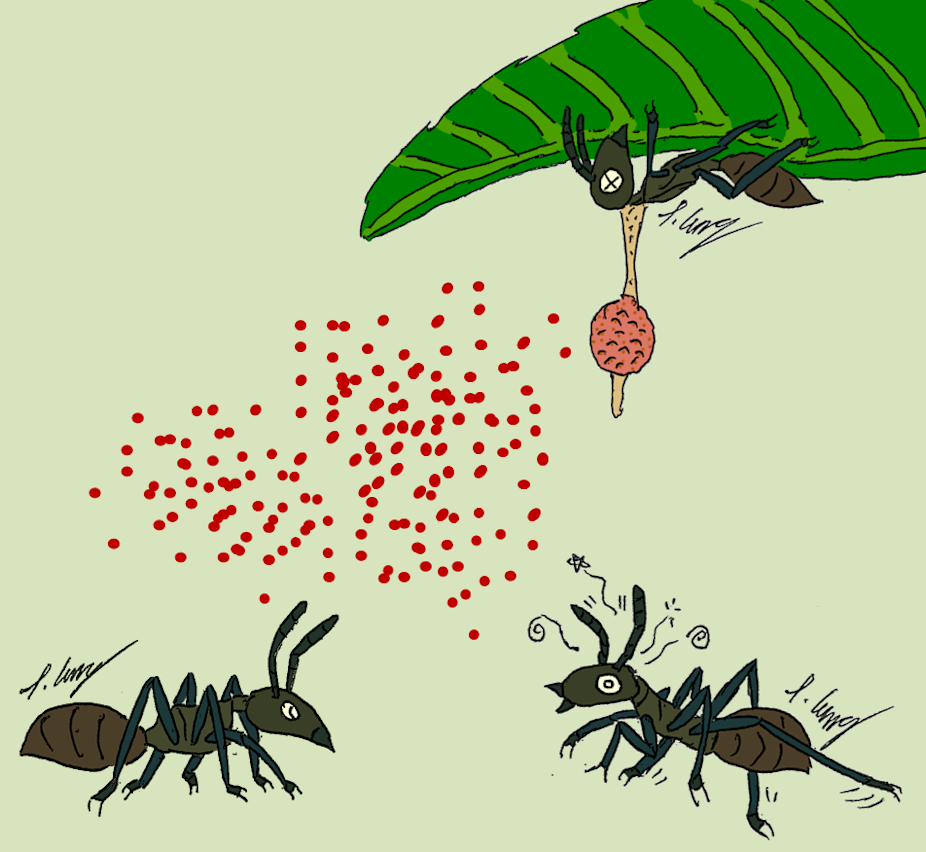
Hyperparasitic fungi can play a vital role in the control of Ophiocordyceps–host interactions, when successfully found in the population (Morozov et al. 2007) were hyperparasitized (Andersen et al. 2012), the much lower proportions of hyperparasitism observed suggest a relatively weak impact on the entomopathogenic.
Good to know
Hyperparasitism: the parasitic habit of one species upon another parasitic species.
Ophiocordyceps species infecting ants – the so-called zombie-ant fungi – incorporate one of the most intriguing and fascinating relationships between microbes and animals. Despite their massive importance for ecosystem functioning, these fungal pathogens are still faintly documented, especially regarding their diversity, ecology, and evolutionary relationships. The natural occurrence in Camponotini ants suggests that this fungus may play a significant role in ant control, and should exhort more widespread studies on the possibility of manipulating this fungus to be used in biological control programs.
References
- (2015) ‘Record of Ophiocordyceps unilateralis sensu lato, the zombie-ant fungus, parasitizing Camponotus in an urban fragment of Atlantic Rainforest in southeastern Brazil.’ Studies on Neotropical Fauna and Environment, 50:1, 21-23.
- Harry C. Evans, Simon L. Elliot, David P. Hughes. (2011) ‘Hidden Diversity Behind the Zombie-Ant Fungus Ophiocordyceps unilateralis: Four New Species Described from Carpenter Ants in Minas Gerais, Brazil.’ PLoS ONE 6(3): e17024.
- (2020) ‘Zombie-ant fungi cross continents: II. Myrmecophilous hymenostilboid species and a novel zombie lineage.’ Mycologia, 112:6, 1138-1170.
- J.P.M. Araújo, H.C. Evans, R. Kepler, D.P. Hughes. (2018) ‘Zombie-ant fungi across continents: 15 new species and new combinations within Ophiocordyceps. I. Myrmecophilous hirsutelloid species.’ Studies in Mycology, 90, 119-160.
- João Carvalho, Letícia Franco de Almeida Costa, Sobczak JF, Salgado-Neto G. (2017) ‘The zombie ants parasitized by the fungi Ophiocordyceps camponoti- atricipis (Hypocreales: Ophiocordycipitaceae): new occurrence and natural history.’ Mycosphere 8 (9): 1261–1266.
- Joa˜ o P.M. Arau´ jo, David P. Hughes. (2019) ‘Zombie-Ant Fungi Emerged from Non-manipulating, Beetle-Infecting Ancestors.’ Current Biology 29 :21, 3735-3738.
- Kai-Wen Tu, Ming-Chung Chiu, Wei-Jiun Lin, Yen-Ping Hsueh, Chung-Chi Lin, Jui-Yu Chou. ‘Bacillus Thuringiensis Predominated in the Zombie Fungus Ophiocordyceps Unilateralis S. L.-Infected Ant Cadavers and Investigation of its Biological Properties.’
- Stephen J. Saltamachiaa, João P. M. Araújob. (2020) ‘Ophiocordyceps desmidiospora, a basal lineage within the “Zombie-Ant Fungi” clade.’ Mycologia, 112:6, 1171-1183.
 Plantlet The Blogging Platform of Department of Botany, University of Dhaka
Plantlet The Blogging Platform of Department of Botany, University of Dhaka






Thanks Again. Cool.
I loved as much as you’ll receive carried out right here. The sketch is attractive, your authored material stylish. nonetheless, you command get bought an nervousness over that you wish be delivering the following. unwell unquestionably come more formerly again as exactly the same nearly a lot often inside case you shield this hike.
Thank you so much for your Valuable comment. We wish to hear from you again.
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
Hi i think that i saw you visited my web site thus i came to Return the favore Im attempting to find things to enhance my siteI suppose its ok to use a few of your ideas
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
I really like reading through a post that can make men and women think. Also, thank you for allowing me to comment!
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
Good post! We will be linking to this particularly great post on our site. Keep up the great writing
You’re so awesome! I don’t believe I have read a single thing like that before. So great to find someone with some original thoughts on this topic. Really.. thank you for starting this up. This website is something that is needed on the internet, someone with a little originality!
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
There is definately a lot to find out about this subject. I like all the points you made
I liked it as much as you did. Even though the picture and writing are good, you’re looking forward to what comes next. If you defend this walk, it will be pretty much the same every time.
I appreciate you sharing this blog post. Thanks Again. Cool.
This is my first brief visit, and I’m delighted to have everything in one location.
Wonderful web site. Lots of useful info here. I’m sending it to a few friends ans additionally sharing in delicious. And obviously, thanks to your effort!
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
Hi my loved one I wish to say that this post is amazing nice written and include approximately all vital infos Id like to peer more posts like this
Fantastic site A lot of helpful info here Im sending it to some buddies ans additionally sharing in delicious And naturally thanks on your sweat
My brother suggested I might like this blog He was totally right This post actually made my day You can not imagine simply how much time I had spent for this info Thanks
I do not even know how I ended up here but I thought this post was great I dont know who you are but definitely youre going to a famous blogger if you arent already Cheers
Usually I do not read article on blogs, however I would like to say that this write-up very compelled me to take a look at and do so! Your writing taste has been amazed me. Thanks, quite nice post.
I’m always amazed at the quality of your writing. This post was no exception. Fantastic job!
Thank you for the good writeup It in fact was a amusement account it Look advanced to far added agreeable from you However how could we communicate
Fantastic read! I was especially impressed by the depth provided on the topic, offering a perspective I hadn’t considered. Your insight adds significant value to the conversation. For future articles, it would be fascinating to explore more to dive deeper into this subject. Could you also clarify more about the topic? It caught my interest, and I’d love to understand more about it. Keep up the excellent work!
Thank you so much for your kind word.. Which clarification do you need?
What i do not understood is in truth how you are not actually a lot more smartlyliked than you may be now You are very intelligent You realize therefore significantly in the case of this topic produced me individually imagine it from numerous numerous angles Its like men and women dont seem to be fascinated until it is one thing to do with Woman gaga Your own stuffs nice All the time care for it up
I’m always impressed by your ability to write about complex topics in an understandable way.
In addition, I found your article to be very well-researched and substantiated. The data and case studies you provided lend credibility to your arguments and demonstrate the potential for success when targeting the right niche. Thank you for putting so much thought and effort into creating this comprehensive resource.
Your composition was remarkably enlightening! The comprehensiveness of the data and the riveting presentation enthralled me. The magnitude of investigation and proficiency apparent throughout substantially elevates the substance’s caliber. The revelations in both the opening and concluding segments were particularly gripping, igniting novel notions and queries that I hope you’ll explore in forthcoming compositions. If there are supplementary resources for further delving into this subject, I’d be delighted to immerse myself in them. Gratitude for sharing your expertise and enriching our comprehension of this topic. The exceptional quality of this piece compelled me to comment promptly after perusing. Maintain the… Read more »
This piece was incredibly enlightening! The level of detail and clarity in the information provided was truly captivating. The extensive research and deep expertise evident in this article are truly impressive, greatly enhancing its overall quality. The insights offered at both the beginning and end were particularly striking, sparking numerous new ideas and questions for further exploration.The way complex topics were broken down into easily understandable segments was highly engaging. The logical flow of information kept me thoroughly engaged from start to finish, making it easy to immerse myself in the subject matter. Should there be any additional resources or… Read more »
This piece was incredibly enlightening! The level of detail and clarity in the information provided was truly captivating. The extensive research and deep expertise evident in this article are truly impressive, greatly enhancing its overall quality. The insights offered at both the beginning and end were particularly striking, sparking numerous new ideas and questions for further exploration.The way complex topics were broken down into easily understandable segments was highly engaging. The logical flow of information kept me thoroughly engaged from start to finish, making it easy to immerse myself in the subject matter. Should there be any additional resources or… Read more »
you are in reality a good webmaster The website loading velocity is amazing It sort of feels that youre doing any distinctive trick Also The contents are masterwork you have done a fantastic job in this topic
Usually I do not read article on blogs, however I would like to say that this write-up very compelled me to take a look at and do it! Your writing style has been amazed me. Thank you, very nice article.
What i do not understood is in truth how you are not actually a lot more smartlyliked than you may be now You are very intelligent You realize therefore significantly in the case of this topic produced me individually imagine it from numerous numerous angles Its like men and women dont seem to be fascinated until it is one thing to do with Woman gaga Your own stuffs nice All the time care for it up
What a remarkable article! The way you’ve tackled the topic with such precision and depth is commendable. Readers are sure to gain a great deal from the wealth of knowledge and practical insights you’ve shared. Your profound understanding of the subject shines through every part of the piece. I’m eager to see more of your exceptional work. Thank you for offering your expertise and providing us with such enlightening and comprehensive content.
What a remarkable article! The way you’ve tackled the topic with such precision and depth is commendable. Readers are sure to gain a great deal from the wealth of knowledge and practical insights you’ve shared. Your profound understanding of the subject shines through every part of the piece. I’m eager to see more of your exceptional work. Thank you for offering your expertise and providing us with such enlightening and comprehensive content.
I simply could not go away your web site prior to suggesting that I really enjoyed the standard info a person supply on your guests Is going to be back incessantly to investigate crosscheck new posts
Fantastic site Lots of helpful information here I am sending it to some friends ans additionally sharing in delicious And of course thanks for your effort