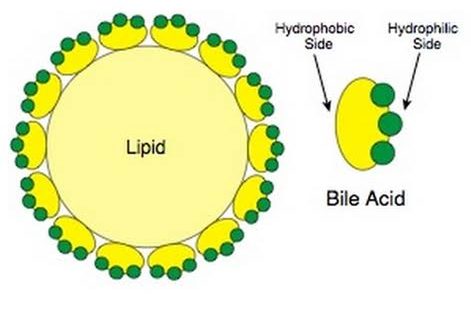
Lipids are a heterogeneous group of compounds that are related to fatty acids. They are usually esters of fatty acids with alcohols. Examples include fats, oils, waxes, and other substances.
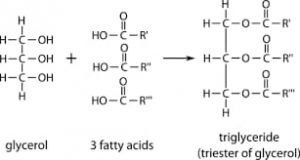
- Fats (solid) and oils (liquid) = Glycerol (tri-hydroxy alcohol) + 3 fatty acids.
- Wax = Mono-hydroxyl alcohol + Fatty acid.
One prominent example of lipid is fat which can also be called triglycerides as they are esters created from glycerol and three fatty acids. 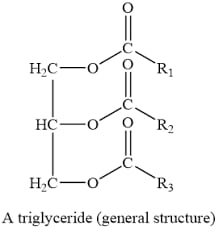
Discovery
The term “lipid” was 1st used by the German biochemist Bloor in 1943.
Occurrence
- Occurs in the cytoplasm as microscopic glucoles or droplets.
- They are widely distributed throughout the plant kingdom.
- They are prevalent in all parts of plants including root, fruit, stem, leaf, flower even in the pollen grain. They are especially abundant in spores and seeds. In general, the vegetative organs contain less than 5% of fat on a weight basis and many seeds about 35-50%.
Characteristics
- Consists of 2 parts: One being alcohol (glycerol) and other fatty acids.
- Insoluble in water as they are non-polar and considerably soluble in non-polar organic solvents like chloroform or benzene. So they are hydrophobic.
- They are colorless and odorless if they are pure. They are capable of absorbing a variety of odors.
- They are storehouses of energy and provide insulation.
- At ordinary room temperature, they may be considered as fats if solid or semi-solid and oils if liquid.
- The melting point of lipids depends on the chain length of the constituent fatty acid and the degrees of unsaturation.
Fats containing saturated fatty acid from Carbon (4-6) are liquid at room temperature, but containing C-10 or higher unsaturated fatty acids are solid and their melting points increase with increasing chain length. - The specific gravity of lipid is less than 1 (about 0.86) and they float on the water surface.
- They are poorer in oxygen in comparison to carbon.
- Usually, fruits or seeds if rich in fats are low in carbohydrates and vice versa.
- Seeds of cereals are not rich in fat. Nuts have the highest oil content but fruits are not high in fat (exception- Olive and avocado).
- Lipids possess high insulating power like they are bad conductors of heat.
Classification
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
Best Money Earning Website 100$ Day
#1 Top ranking article submission website
The following classification of lipids has been proposed by Bloor on the basis of their chemical composition.
1. Simple lipid
2. Compound lipid
3. Derived lipid
Simple lipid
It is also called homolipid. Simple lipid is esters of acids with various alcohol. There are 2 types of simple lipids.
a) Triglycerides (fats and oils)
- They are esters of fatty acids with a tri-hydroxyl alcohol and they are the simplest fat.
- Insoluble in water (hydrophobic and nonpolar).
- A storehouse of energy.
- Present in many plants, mainly in seeds.
The main constituents of vegetable oil (typically mono unsaturated) and animal fat (typically mono saturated).
b) Waxes
Waxes are fatty acids with high molecular weight mono hydroxyl alcohol.
Compound lipid
They can be called heterolipids. They are esters of fatty acid with alcohol with additional groups also. Example- Phospholipid, glycolipid etc.
a) Phospholipid
Phospholipids are the lipids containing fatty acids and glycerol with a phosphonic nitrogen bases and other substituents.
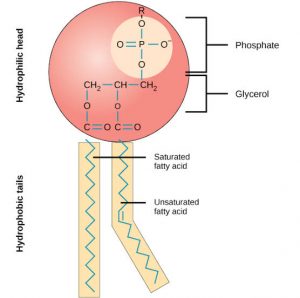
- It is a very important structural component for cell membrane.
- The structure of phospholipids molecules generally consists of a hydrophobic tail and a hydrophilic head.
The tail consists of hydrocarbon chains whereas phosphate and amino groups are present in the head of phospolipid. - It can be divided into 3 groups- Phospho-glycerides, phospho-inositides, Phospho-phinogosides.
There are numerous types of phospholipid in nature. Among them lecithin and cephalin are very common. Lecithins are widely distributed in nature. It is found in various oil seeds like- soybeans.
b) Glycolipid
It is also called galactolipid, cerebrosides (it is a glycolipid that is found primarily in the brain and peripheral nervous tissue).
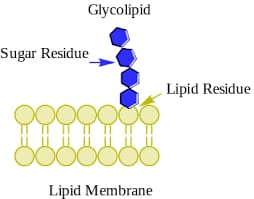
- Composed of one membrane compounds and covalently bonded to monosaccharides.
- Are the compounds of fatty acids with carbohydrates and contain nitrogen but no phosphoric acid.
- Provide energy and also serve as markers of cellular recognition.
- Example- Gangliosides, sulfolipids and sulfatids
Derived lipid
Derived lipids are produced when the hydrolysis of simple or compound lipids take place. Example – Steroids, terpens, monoglycerides, diglycerides, alcohol etc.
Biological Significance of lipids
- Lipid serves as a source of energy.
- They are involved in the building of cellular compounds like – phospolipids are present in biological membrane.
- In animals, fat often deposits under the skin and protects against extreme environmental conditions.
- Fatty tissues around many vital organs protect against mechanical injury also.
- They act as co-factors and hormones.
- They also form the protective covering (wax) in plants, animals.
Undoubtedly, lipid plays a very important role in living organisms by energy provision, maintenance of temperature, formation of lipid bi-layer membrane, chemical messengers and so on.
Some scientific papers on this topic
- Free: Biochemistry of lipid metabolism in olive and other oil fruits. Salas, Joaquı́n J., et al. “Biochemistry of lipid metabolism in olive and other oil fruits.” Progress in Lipid Research 39.2 (2000): 151-180.
- Free: Lipid biosynthesis. Ohlrogge, John, and John Browse. “Lipid biosynthesis.” The Plant Cell 7.7 (1995): 957.
- Log in: Lipid oxidation. Frankel, E. N. “Lipid oxidation.” Progress in lipid research 19.1-2 (1980): 1-22.
- Log in: Structure of lipid bilayers. Nagle, John F., and Stephanie Tristram-Nagle. “Structure of lipid bilayers.” Biochimica et Biophysica Acta (BBA)-Reviews on Biomembranes 1469.3 (2000): 159-195.
Revisions
- Partially revised by Abulais Shomrat on 19 October, 2020.
- Get Free Netflix Now
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
 Plantlet The Blogging Platform of Department of Botany, University of Dhaka
Plantlet The Blogging Platform of Department of Botany, University of Dhaka