[Click here to read ‘Bichemical Cycle (part- 1 )’ ]
Threats to the Carbon Cycle
- Decline in forest growth.
- Killing of ocean phytoplankton due to rising sea temperatures.
- Death of forests due to spread of disease and insects.
- Melting permafrost layer (In geology, permafrost or permafrost soil is soil at or below the freezing point of water for two or more years. Permafrost can also be a storage of carbon (1.7 billionMT).
- Land clearing for development and agriculture.
- Continued output of carbon from fossil fuel burning.
Human induced changes in the global carbon cycle
– The Earth is getting warmer.
– The 20th century was the warmest in the last 600 years.
– This century is about 1 degree Fahrenheit warmer than last century.
-The balance of evidence suggests that burning of fossil fuel(e.g. coal, oil, natural gas), which emits CO2 as a waste, is the cause.
– CO2 is a “Green House” gas. It traps heat at the Earth’s surface.
Sedimentary Cycles
Phosphorus (P) Cycle
P is a constituent element of the organic compounds including phosphorylated sugars (e.g. fructose phosphate), phospholipids, nucleotides (e.g. ATP) and nucleic acids (e.g. DNA, RNA). It is a highly immobile element and it moves in a sedimentary cycle.
- Its transformation in the geochemical cycle is simpler than the N cycle.
- The source of P is rock. It is released into the cycle through erosion or mining.
- It is soluble in H2O as phosphate (PO4)
- It is taken up by plant roots, then travels through food chains.
- Following the death of plants and animals their decomposition by micro-organisms releases P in the from of phosphates.
The following picture is a simplified model of the circulation of various chemical forms of phosphorus (mostly phosphates) in the phosphorus cycle, with major harmful human impacts shown by the red arrows.
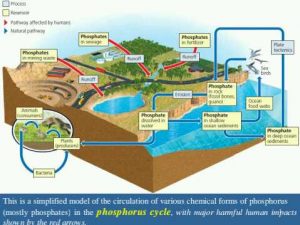
- In contrast to the cycles of water, carbon, and nitrogen, the phosphorus cycle does not include the atmosphere.
- The major reservoir for P is phosphate salts containing phosphate ions (PO43–) in terrestrial rock formations and ocean bottom sediments.
- The P cycle is slow compared to the water, carbon, and10nitrogen cycles.
- Phosphate can be lost from the cycle for long periods of time when it is washed from the land into streams and rivers and is carried to the ocean.
- There it can be deposited as marine sediment and remain trapped for millions of years.
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
Best Money Earning Website 100$ Day
#1 Top ranking article submission website
Someday, geological processes may uplift and expose these seafloor deposits, from which phosphate can be eroded to start the cycle again.
Affect of Human activities
This includes removing large amounts of phosphate from the earth to make fertilizer and reducing phosphate levels in tropical soils by clearing forests.
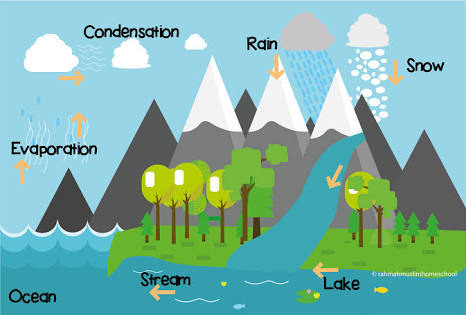
Hydrologic cycle: Water Cycle
Water is one of the most imprtant resources on earth. It has a critical function in all most all sphere of life.
It occurs in 3 states of matter-
- solid sate (e.g. ice),
- liquid state (e.g. water) and
- gaseous stage (e.g. vapour).
A part of water is in continuous circulation because it evaporates from water and land surfaces of the earth.
After condensation in the atmosphere it is deposited again on earth surface as solid or liquid precipitation.
- By comparing with the mean annual precipitation on the earth(97 cm), it follows that average life of water molecule in the atmosphere is about 10 days.
Thus, water cycle involves evaporation, condensation and precipitation.
- About 97% of water is oceanic and saline
- 1.74% is glaciers and ice caps.
- Shallow ground water is 0.76%.
The usable water on surface of the earth, on which mankind and economy and industry are dependent constitutes only a minute fraction of the total water of the earth and is supplied by water cycle.
 Plantlet The Blogging Platform of Department of Botany, University of Dhaka
Plantlet The Blogging Platform of Department of Botany, University of Dhaka