A disaccharide, also called double sugar, is the sugar formed when two monosaccharides (simple sugars) are joined by a glycosidic linkage. Like monosaccharides, disaccharides are soluble in water. Three common examples are sucrose, lactose, and maltose.
Disaccharides are one of the four chemical groupings of carbohydrates (monosaccharides, disaccharides, oligosaccharides, and polysaccharides). The most common types of disaccharides—sucrose, lactose, and maltose—have 12 carbon atoms, with the general formula C12H22O11. The differences in these isomeric disaccharides are due to atomic arrangements within the molecule.
Glycosidic linkage
Glycosidic bond is a covalent chemical bond that holds together a glycoside. A glycoside is simply a ring-shaped sugar molecule that is attached to another molecule with an -OR group.
The sugar ring may be either a 5-membered ring or a 6-membered ring, and the other molecule can be a non-sugar or a sugar one, but in case of dissacharides, this other molecule is always a sugar (can be 5-membered ring or a 6-membered ring).
Sucrose
Sucrose, being a disaccharide, is composed of two sugar units, a glucose and a fructose, linked by a glycosidic bond. It is also known as cane sugar, table sugar or beet sugar.
Properties of sucrose
- General formula of sucrose is C12H22O11.
- It occurs as a colorless crystalline form.
- It is soluble in water and have a melting point of 160 degree celcius.
- There is no free aldehyde and ketone group. So it is a non reducing sugar.
- It does not behave as a reducing agent towards oxidizing metal salts. It doesn’t give any color while reacting with ‘Fehling’s solution’.
- It occurs in various amount in different plant organs such as fruits, seeds, flowers, roots etc.
- It is the chief element of sugar cane, beet, maple, pineapple and carrot.
- Mostly found in nectar of flowers. It is the raw material of honey.
- It is two times sweeter than glucose.
Formation of sucrose
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
Best Money Earning Website 100$ Day
#1 Top ranking article submission website
In the sucrose molecule, two rings are present
- Pyranose ring where glucose is present as a 6 carbon ring.
- Furanose ring where fructose is present as 5 carbon ring.
Sucrose, in a nutshell
- Sucrose = Glucose + Fructose
- Glucose type: alpha, D
- Fructose type: beta, D
- Linkage: 1, 2 glycosidic linkage (meaning the 1-C of glucose is linked with 2-C of fructose)
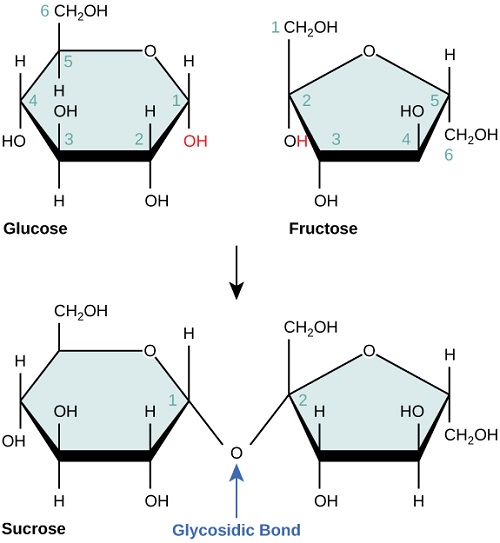
(Here, fructose is inverted to bring the 2-C near the 1-C of the glucose to form a glycosidic bond)
Importance of sucrose
- It is used as a sweeter in foods and soft drinks, in the manufacture of syrups, confectionary, pharmaceutical products, caramel etc.
- Translocation of food takes place mainly in the form of sucrose. It is synthesized during photosynthesis.
- The annual world production of sucrose is about 90 million tons. Mainly produced from the juice of sugar cane (20%) and sugar beet (17%).
- It is used in food products as a preservative, antioxidant and moisture control agent.
Lactose
The name lactose comes from lac (Latin ‘lactis’ meaning milk) and ‘ose’ is used to indicate sugars. It is composed of one molecule of galactose and one molecule of glucose.
- It makes up around 2-8% milk.
- It is white, water soluble, solid with a mildy sweet taste.
- This sugar is made up by Beta-1,4 linkage.
Formation of Lactose
Lactose, in a nutshell
- Lactose = Galactose + Glucose
- Galactose type: beta, D
- Glucose type: beta, D
- Linkage: 1, 4 glycosidic linkage (meaning the 1-C of Galactoseis linked with 4-C of Glucose)
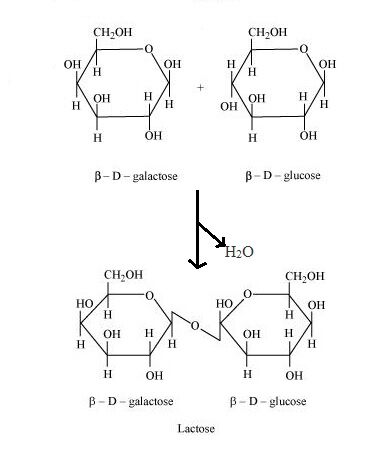
(Here, no inversion of any molecule is required.)
Importance of lactose
- Lactose aids the absorption of calcium and phosphorous.
- It supports the growth of friendly bacteria in human intestine. The growth of friendly bacteria helps promoting a healthy digestive system.
- Galactose, the simple sugar found in lactose carries a role for the development of the brain and nervous system.
- Lactose, in breast milk, is a natural and sole source of carbohydrate which provides energy.
- Lactose in mammal’s milk is very important for the development of infants.
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
 Plantlet The Blogging Platform of Department of Botany, University of Dhaka
Plantlet The Blogging Platform of Department of Botany, University of Dhaka





