The word protein refers to a type of molecule in food that can be broken down into amino acids. They are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form, facilitating a biological function. Protein accounts on average about 1/4th to 1/3rd of the total dry weight of any living organisms. They are considered the working molecules of the cells.
Discovery
Proteins were first described by the Dutch chemist Geradus Johannes Muller and named by Swedish chemist Jöns Jacob Berzelius in 1838.
Characteristics of Protein
- Proteins are high molecular weight compounds. They have a molecular weight of 5 to 300 kilodaltons (1 kilo Dalton = 1.67 * 10-21 grams).
- They are made of several amino acids. Typically 20 different amino acids synthesize all the necessary proteins. Note that, a protein doesn’t necessarily require all of the 20 amino acids for its formation.
- The backbone of all protein molecules is made up of a linear chain of polypeptides, which are made up of amino acids.
- Proteins are colorless and tasteless (except serine which is sweet).
- The number of amino acids in the polypeptide may vary considerably. For example, The smallest protein ‘insulin’ has 51 amino acids and the bigger protein ‘hemoglobin’ has 574 amino acids.
- The union between amino acids involves amino(-NH2) and carboxyl (-COOH) groups and releases water. Such bonds (=CO-NH) are called peptide bonds.
- Every peptide has N-terminus and C-terminus. N-terminus is one end of the polypeptide, where the free amino group is present, at the other end the C-terminus free carboxyl group is present.
- Plants are able to synthesize all these amino acids from breakdown products of carbohydrates and reduced N.
- Range in shape from simple crystalloid spherical structures to long fibrillar structures.
- Proteins are the fundamental constituent of the cytoplasm of cells.
- Proteins are considered bricks, they make up bones, muscles, hairs, and other parts of the body.
- The diffusion rate of protein is extremely low.
- Proteins exhibit amphoteric properties i.e. they can act as acids and alkalies.
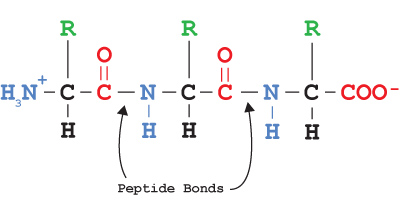
Peptide bond of protein molecule. This happens due to the presence of carboxyl and amino groups in the amino acids of the protein molecule. Though these groups are used in peptide bond formation between each two adjacent amino acids during making a polypeptide chain, the long chain has an open carboxyl group at one end (rightmost group in the figure) and an amine group at another end (leftmost group in the figure).
- Protein-like enzymes are functional elements that take part in metabolic reactions.
- Antibodies and blood hemoglobin are also made of protein.
- Peptides are the simplest protein.
Structures of Protein
Depending on the structure of the proteins, they are classified into four groups named as:
- Primary structure
- Secondary structure
- Tertiary structure
- Quaternary structure
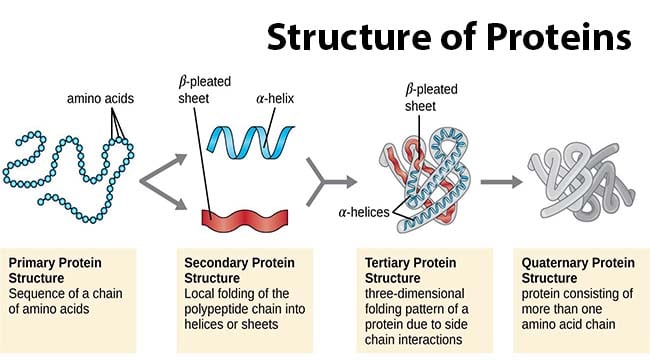
1. Primary structure
Primary structure describes the unique order in which amino acids are linked together to form a protein. Proteins are constructed from a set of 20 amino acids.
2. Secondary structure
Secondary structure refers to the coiling or folding of a polypeptide chain that gives the protein its 3-D shape. There are two types of secondary structures.
- The alpha helix structure
- The beta pleated sheet
3. Tertiary structure
Tertiary structure refers to the comprehensive 3-D structure of the polypeptide chain of a protein. There are several types of bonds and forces that hold it in its tertiary structure. These are
- Hydrophilic structure
- Hydrogen bonding
- Ionic bonding
4. Quaternary structure
Quaternary structure refers to the structure of a protein macromolecule formed by interactions between multiple polypeptide chains. Each polypeptide chain is referred to as a subunit. Proteins with quaternary structures may consist of more than one of the same or different subunits. Example: Hemoglobin.
 Plantlet The Blogging Platform of Department of Botany, University of Dhaka
Plantlet The Blogging Platform of Department of Botany, University of Dhaka






It’s really fabulous to understand regarding protein.
This article describes best of protein from my opinion. Thanks for sharing