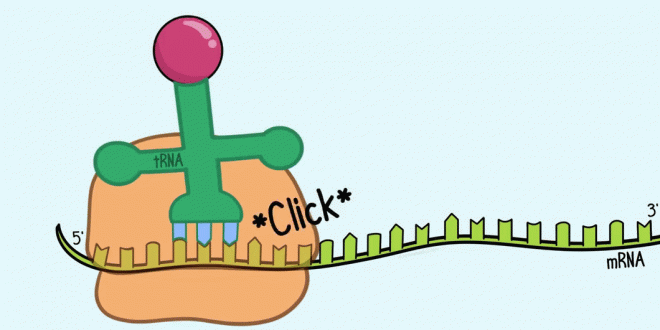
Translation takes place on ribosomes; indeed, ribosomes can be thought of as moving protein-synthesizing machines. A ribosome attaches near the 5′ end of an mRNA strand and moves toward the 3′ end, translating the codons as it goes.
Synthesis begins at the amino end of the protein, and the protein is elongated by the addition of new amino acids to the carboxyl end of the former amino acid.
Requirements
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
Best Money Earning Website 100$ Day
#1 Top ranking article submission website
1. mRNA
Acts as the template for proten synthesis.
2. Ribosome
Ribosome is the platform where tRNA sits. It has two subunits: one smaller and another one greater. The sub-units usually remain separately suspended in the cytoplasm but joins together when translation commences. It has three sites primarily know as P site, A site and E site.
3. A set of three proteins called initiation factors
4. tRNA with Amino Acids
There are 20 types of Amino Acids (AAs) that code for the proteins in every organisms body. These AAs are carried by tRNAs to the ribosome-mRNA complex. For 20 types of Amino acids, there are 61 codons.
5. Guanosine triphosphate (GTP)
To supply with the energy in this complex process.
6. Activating enzymes
This enzymes helps in Amino Acid activation.
Protein synthesis can be conveniently divided into four stages:
- The binding of amino acids to the tRNAs.
- Initiation: The components necessary for translation are assembled at the ribosome.
- Elongation: The amino acids are joined, one at a time, to the growing polypeptide chain.
- Termination: The protein synthesis halts at the termination codon and the translation components are released from the ribosome.
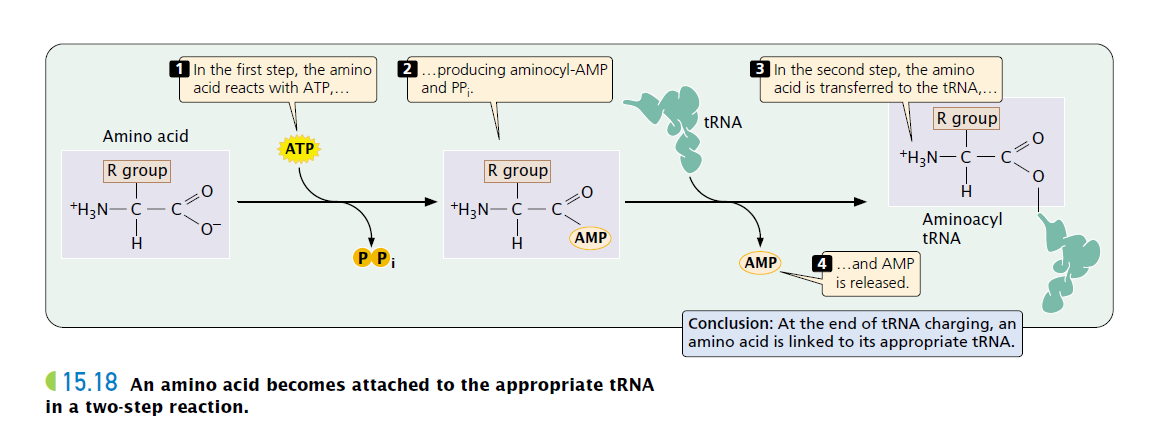
Amino Acid Activation
The first stage of translation is the binding of tRNA molecules to their appropriate amino acids. When linked to its amino acid, a tRNA delivers that amino acid to the ribosome, where the tRNA’s anticodon pairs with a codon on mRNA. This process enables the amino acids to be joined in the order specified by the mRNA. Proper translation, then, first requires the correct binding of tRNA and amino acid.
- A cell typically possesses from 30 to 50 different tRNAs, and, collectively, these tRNAs are attached to the 20 different amino acids.
Each tRNA is specific for a particular kind of amino acid. All tRNAs have the sequence CCA at the 3′ end, and the carboxyl group (COO-) of the amino acid is attached to the 2- or 3- hydroxyl group of the adenine nucleotide at the end of the tRNA.
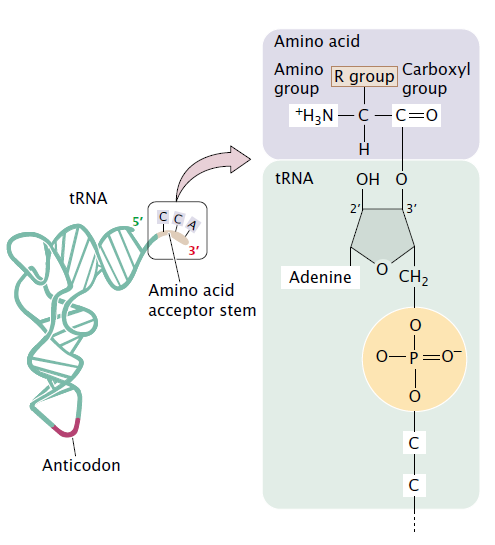
- Errors in tRNA charging are rare; they occur in only about 1 in 10,000 to 1 in 100,000 reactions.
- Activating enzymes, aka Amino-Acyl tRNA synthatase helps to complete this process.
Initiation
During initiation, all the components necessary for protein synthesis assemble.
- tRNA containing Amino acid formalmethionine (only methionine in eukaryotes). The complex is represented as tRNAfMet.
- Two ribosomal sub-units.
- Initiation complex.
- GTP for energy.
Initiation comprises three major steps.
- First, mRNA binds to the small subunit of the ribosome.
- Second, initiator tRNA binds to the mRNA through base pairing between the codon and anticodon.
- Third, the large ribosome joins the initiation complex.
The sequence covered by the ribosome during initiation is from 30 to 40 nucleotides long and includes the AUG initiation codon. Then at first a 30S initiation complex is formed and then by adding the large sub-unit and by subsequent dislocation of some initiation factors, form 70S initiation complex (in prokaryotes).
Elongation
The next stage in protein synthesis is elongation, in which amino acids are joined to create a polypeptide chain. Elongation requires (1) the 70S complex just described; (2) tRNAs charged with their amino acids; (3) several elongation factors (EF-Ts, EF-Tu, and EF-G); and (4) GTP.
A ribosome has three sites that can be occupied by tRNAs; the aminoacyl, or A, site, the peptidyl, or P, site, and the exit, or E site.
The initiator tRNA immediately occupies the P site (the only site to which the fMet-tRNAfMet is capable of binding), but all other tRNAs first enter the A site. After initiation, the ribosome is attached to the mRNA, and fMet-tRNAfMet is positioned over the AUG start codon in the P site; the adjacent A site is unoccupied.
Elongation occurs in three steps. The first step is the delivery of a charged tRNA (tRNA with its amino acid attached) to the A site.
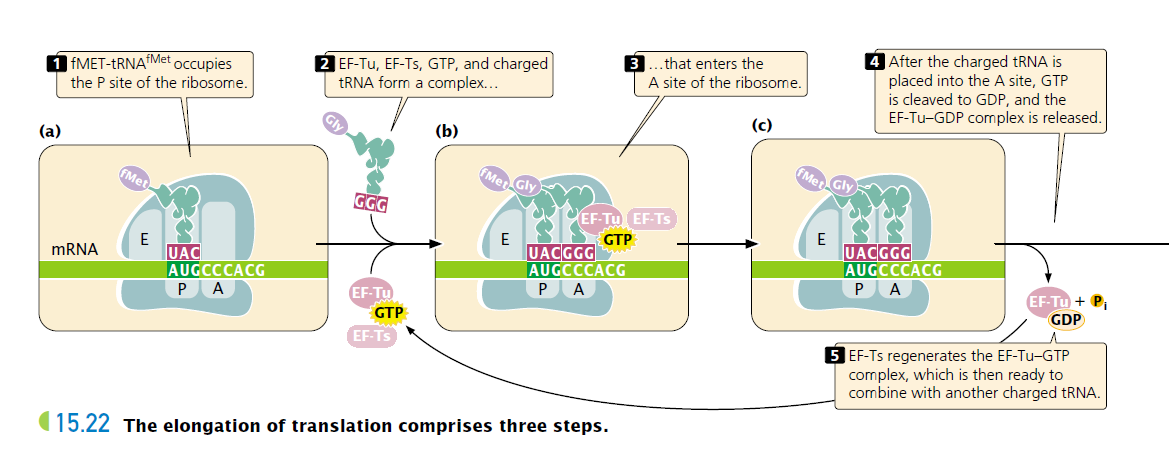
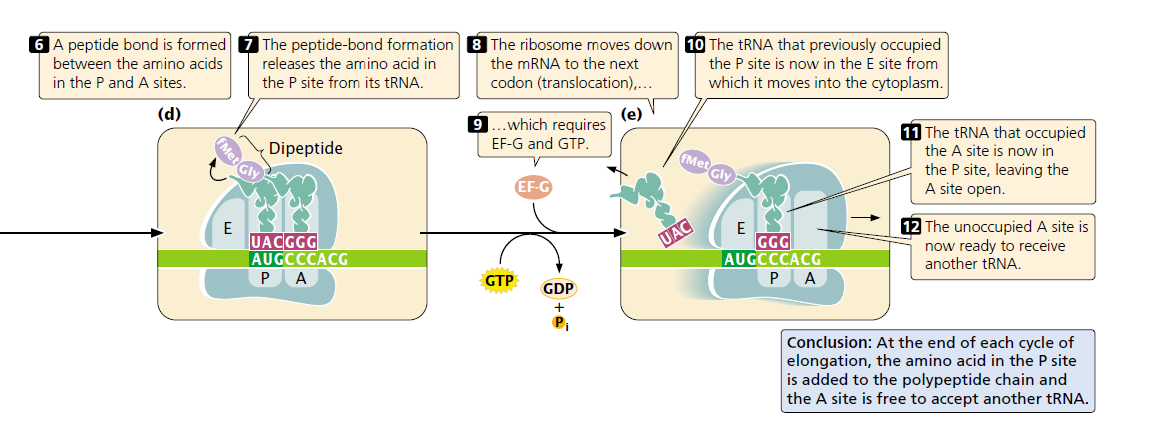
The second step of elongation is the creation of a peptide bond between the amino acids that are attached to tRNAs in the P and A sites ( FIGURE 15.22d). The formation of this peptide bond releases the amino acid in the P site from its tRNA.
The third step in elongation is translocation the movement of the ribosome down the mRNA in the 5′ to 3′ direction. Because the tRNAs in the P and A site are still attached to the mRNA through codon– anticodon pairing, they do not move with the ribosome as it translocates. Consequently, the ribosome shifts so that the tRNA that previously occupied the P site now occupies the E site, from which it moves into the cytoplasm where it may be recharged with another amino acid.
Translocation also causes the tRNA that occupied the A site (which is attached to the growing polypeptide chain) to be in the P site, leaving the A site open. Thus, the progress of each tRNA through the ribosome during elongation can be summarized as follows: cytoplasm → A site → P site → E site → cytoplasm. After translocation, the A site of the ribosome is empty and ready to receive the tRNA specified by the next codon. The elongation cycle repeats itself:
Termination
Protein synthesis terminates when the ribosome translocates to a termination codon. Because there are no tRNAs with anticodons complementary to the termination codons, no tRNA enters the A site of the ribosome when a termination codon is encountered. Instead, proteins called release factors bind to the ribosome.
The release factors then promote the cleavage of the tRNA in the P site from the polypeptide chain.
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
 Plantlet The Blogging Platform of Department of Botany, University of Dhaka
Plantlet The Blogging Platform of Department of Botany, University of Dhaka