Introduction
Plant breeding has been practised for thousands of years, since the rudiments of human civilization. It is now discussed worldwide by government institutions.
Plant breeding is the purposeful manipulation of plant science in order to create desired genotypes and phenotypes for quest. This manipulation involves either controlled pollination, genetic engineering or both followed by a artificial selection for progeny.
 Classical plant breeding was extremely successful in generating high yielding crop varieties. Yet in modern crops, the domestication process impoverished the genetic diversity available for breeding. This is limiting further improvements of elite germplasm by elegant approaches. And also under pressure of rapid population growth, climate change and agricultural pests and diseases, the green evolution with new technologies are required to address and provide novel genetic variations to improve yield, quality and resistance against biotic and abiotic stresses in crop plants.
Classical plant breeding was extremely successful in generating high yielding crop varieties. Yet in modern crops, the domestication process impoverished the genetic diversity available for breeding. This is limiting further improvements of elite germplasm by elegant approaches. And also under pressure of rapid population growth, climate change and agricultural pests and diseases, the green evolution with new technologies are required to address and provide novel genetic variations to improve yield, quality and resistance against biotic and abiotic stresses in crop plants.
Already, the most powerful and advanced genome editing system has been discovered, which is known as “CRISPR-Cas9″ system. The CRISPR-Cas9 technology is revolutionizing genome engineering and equipping scientists & breeders with the ability to precisely modify the DNA of crop plants.
As CRISPR-Cas9 technology is the most advanced and impactful for plant breeding, it is necessary for a plant breeder to know about it and how it works.
In this article, a little bit fact about CRISPR CAS-9 technology and how this technology is used on plant breeding will be narrated briefly.
CRISPR-Cas9 Technology
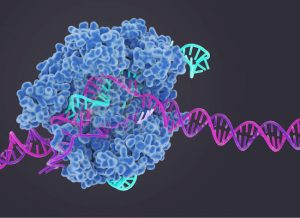
CRISPR stands for Clustererd Regularly Interspace Short Palindromic Repeats which is a family of DNA sequence found in the genome of prokaryotic organisms such as bacteria and archaea.
And Cas9 (or CRISPR associated protien 9) is an enzyme that uses CRISPR sequence as a guide to recognize and cleave specific strands for DNA that are complementary to the CRISPR sequence. Cas9 enzymes together with CRISPR sequences from the basis of a technology known as CRISPR-Cas9 technology that can be used to edit genes within organisms.
Best safe and secure cloud storage with password protection
Get Envato Elements, Prime Video, Hotstar and Netflix For Free
Best Money Earning Website 100$ Day
#1 Top ranking article submission website
Discovery
CRISPR was first discovered in archaea (later in bacteria) by Francisco Mojica, a scientist at the University of Alicante in Spain. He proposd that CRISPR serve as part of the bacterial immune system, defending against invading viruses. They consists of repeating sequences in genetic code, interrupted by “spacer” sequences (remnants of genetic code from past invaders). The system serves as a genetic memory that helps the cell detect and destroy invaders (called bacteriophase) when they return.
Mojica’s theory was experimentally demonstrated in 2007 by a team of scientists led by Philippe Horvath. It was developed in June, 2012 by Emmanuelle Charpentier and Jenifer Doudna, University of California, Berkeley.
In January 2013, the Zang lab published the first method to engineer CRISPR to edit the genome in mouse and human cells. CRISPR-Cas9 has received a lot of attention in recent years due to its range of applications including biological research, breeding and development of agricultural crops and animals and human health applications.


How CRISPR-Cas9 works?
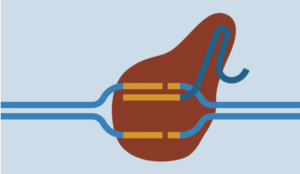
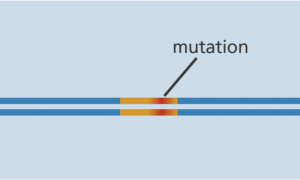
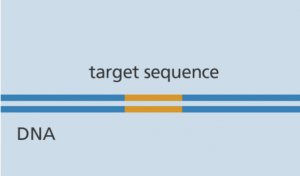 The CRISPR-Cas9 system consists of two key molecules that introduce a change (mutation) into the DNA. These are:
The CRISPR-Cas9 system consists of two key molecules that introduce a change (mutation) into the DNA. These are:
An enzyme called Cas-9. This acts as a pair of “molecular scissor” that can cut the two strands of DNA at a specific location in the genome so that bits of DNA can then be added or removed.
- A piece of RNA called guide RNA (gRNA). This consists of a small piece of pre-designed RNA sequence (about 20
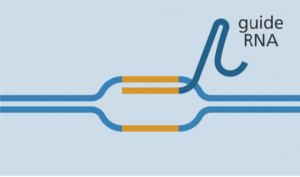
Guide RNA binds to target sequence bases long) located within a longer RNA scaffold. The scaffold part binds to DNA and the pre-designed sequence ‘guides’ Cas-9 to the right part of the genome. This makes sure that the Cas-9 enzyme cuts at the right point in the genome.
- The guide RNA is designed to find and bind to a specific sequence in the DNA. The guide RNA has RNA bases that are complementary to those of the target DNA sequence in the genome. This means that, at least in theory, the guide RNA will only bind to the target sequence and no other regions of the genome.
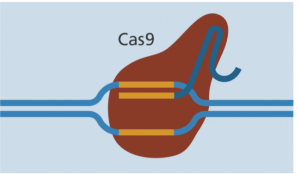
Cas-9 enzyme joins at the same location. - The Cas-9 follows the guide RNA to the same location in the DNA sequence and makes a cut across both strands of the DNA.
- At this stage the cell recognizes that the DNA is damaged and tries to repair it.
- Scientists can use the DNA repair machinery to introduce changes to one or more genes in the genome of a cell of interest.
Application and Recent Advances of CRISPR-Cas9 in Plant Breeding
Today, the application of the CRISPR-Cas9 system-based genome editing for crop improvement has already begun its movement despite still being in its infancy. As a matter of fact, there are already numerous publications that reported that this technology has been successfully implemented in a broad range of plant species. (As shown in table)
Table-1: List of CRISPR-Cas9 system based genome-edited plants
| Species Name | Target Gene (S) | Gene Function | Description | Mode of Action |
| Arabidopsis thaliana | BR11, JAZ1, GAI | Growth regulators | Transgenic plants displayed retarded growth after being subjected to targeted mutagenesis. | Gene disruption |
| Brassica oleracea | BolC.GA4.a | Gibberelin biosynthesis | Transgenic plants displayed dwarf phenotype after being subjected to targeted mutgenesis | Gene disruption |
| Citrus sinensis | CsPDS | Carotinoid biosynthesis | Transgenic plants displayed albinism expression after being subjected to mutagenesis | Gene regulation |
| Cucumis saItivus | eIF4E | Translation initiation factor | Transgenic plants developed resistance towards a broad range of virus | Gene disruption |
| Glycine max | Bar, GmFE11,
GmFE12 |
Root hair growth factors | Transgenic plants displayed higher root hair growth induction after being subjected to targeted mutagenesis | Gene regulation |
| Hordeum vulgare | HvPM19 | Grain dormancy regulator | Transgenic plants displayed signs of dormancy after being subjected to targeted mutagenesis | Gene disruption |
| Marchantia poymorpha | ARF1 | Auxin response
factor |
Transgenic plants showed no response towards auxin after being subjected to targeted mutagenesis | Gene disruption |
| Nicotiana benthamiana | NbPDS | Carotenoid biosynthesis | Transgenic plants displayed albinism expression after being subjected totargeted mutagenesis. | Gene insertion |
| Zea mays | ZmIPK | Phytic acid biosynthetic pathway catalyst | after being subjected Transgenic plants showed reduction of phytic acid level after after being subjected totargeted mutagenesis. | Gene disruption |
| Oryza sativa | OsPDS,OsMPK2,
OsBADH2 etc |
Carotenoid
Biosynthesis growth regulator |
Transgenic plants displayed albinism expression and dwarfism after being subjected to targeted mutagenesis | Gene disruption |
| Petunia hybrid | PDS | Carotenoid biosynthesis | Transgenic plants displayed albinism expression and after being subjected to targeted mutagenesis | Gene disruption |
| Triticum aestivum | TaINOX,
TaPDS |
Inositol mrtabolism and carotenoid biosynthesis | Transgenic plants displayed albinism expression after being subjected to targeted mutagenesis | Gene disruption |
| Solanum lypercosum | SIAGO7 | Involved in RNA biogenesis regulation | Transgenic plants dispayed needle like or lacking lamina leaves after being subjected to targeted mutagenesis | Gene disruption |
| Solanum tuberosum | StALS1 | Acetolactate biosynthesis | Transgenic plant showed increased resistance on herbicides after being subjected to targeted mutagenesis | Gene insertation |
| Sorghum bicolor | DsRED2 | Fluorescence | Transgenic plant showed signs of red fluorescence after being subjected to targeted mutagenesis | Gene disruption |
CRISPR-Cas9 System Based Genome Edited Plants
As the aforementioned plants are successfully genetically modified in the lab but there are actually some of them that are almost readily gainable in the commercial market. These plants may be recent to the market but it is indisputable that they will eventually be susceptible to monopolize the market as they have much more improved traits compared to their relative wild-type plants. As shown in Table-2, most of the plants such as the Wheat and Ranger Russet potato are momentous food staples in many parts of the world and this makes out that the CRISPR-Cas9 system-based genome editing for crop improvement is definitely on its way to revolutionizing the agriculture industry.
TABLE-2: List of CRISPR-Cas9 system-based genome edited plants that are making their way to the commercial market:
| Crops | Traits Improved | Status | Name of Organization |
| White button mushroom | Brownin resistant | Submitting for review to Food and Drug Administration (FDA) | Yinong Yang; Penn State College of Agriculturul Science |
| Waxy corn | Diseases resistant
Drought tolerance |
To be marked within
5-10 years , pending field trials and applicable regulatory reveiw |
DuPont Pioneer |
| Wheat | Produce gluten-free wheat by eliminating
Gliadins in wheat |
Working with gliadin genes that are still present | Institute for Sustainable
Agriculture |
| Soybean | Produce healthier oil with reduced unsaturated fat content by increasing the percentage of oleic acid | Inactivation of two genes in soybean | Institute for Basic Research (IBS) |
| Ranger Russet Potato | Longer freshness because it does not accumulate sweet sugars at typical cold storage tempareture does not ptoduce acylamide (caracinogen ) when fried. | To be grown and sold
In 2019 |
Dan Voytas; Cellectis Plant Science |
With all the studies done so far, it is indisputable that the CRISPR-Cas9 system is on its way to change the pace and courses in the agriculture industry. Perfect plants that have high yield, quality and resistance toward any disease and pests will no longer be impossible with the dawn of this technology. Moreover, CRISPR-Cas9 based gene editing for plants will also be developed to the point where it can be used to replace any defective gene with a normal allele at its natural location. Consequently, all plants will now no longer need to be in danger from any traditional diseases as long as this technique is present and approved for human consumption.
There are still many uncertainities on the usage of plant genome editing. Therefore, in-depth studies are required to ensure this technology will have zero risks while gaining maximum benefits. Besides that, the idea of genome editing might also raise ethical questions from public; these need to be adequately addressed by researches and scientists that are well adept in genome engineering. Educational talk or workshop on genome editing should be given to non-scientists to ensure they understand the basics and benefits of this technology. More or laws and regulations will also be required for the implementation to ensure CRISPR-Cas9 system is well understood and regulated, it will be feasible for the application of this technology to be the maximized to its fullest potential to achieve previously envisioned ideas in plant science.
To recapitulate, we can utter with one voice that CRISPR-Cas9 technology is apparently a benison for plant breeding.
1. http//www.intechopen.com/books/next-generation-plant-breeding/the-crispr-cas9-system-for-crop-improvement-progress-and-prospects
2. https://www.yourgenome.org/facts/what-is-crispr-cas9
3. Source of featured image: https://geneticliteracyproject.org/2018/04/02/whats-stopping-us-from-using-crispr-to-gene-edit-humans-to-fight-disease/
Written by
Mahmuda Hossain Mim, 2nd Year, Dept. of Botany, University of Dhaka
 Plantlet The Blogging Platform of Department of Botany, University of Dhaka
Plantlet The Blogging Platform of Department of Botany, University of Dhaka





Your article helped me a lot, is there any more related content? Thanks!